
(iv) An aqueous solution of CuCl 2 with platinum electrodes. (iii) A dilute solution of H 2SO 4with platinum electrodes. (ii) An aqueous solution of AgNO 3with platinum electrodes. 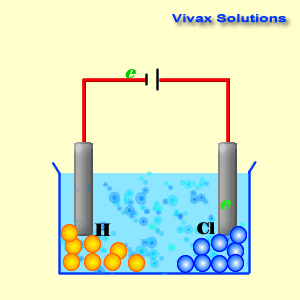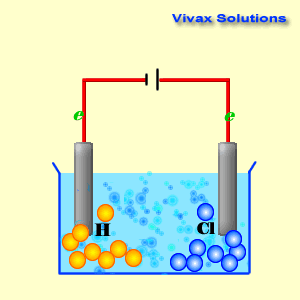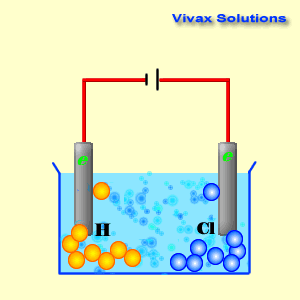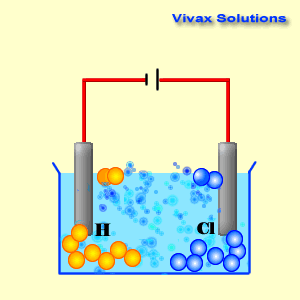
electrolysis in each of the following:(i) An aqueous solution of AgNO3 with. Question 18: Predict the products of electrolysis in each of the following: (i) An aqueous solution of AgNO 3 with silver electrodes. The reaction with a higher value of takes place at the cathode. The positive copper(II) ions Cu 2+ (from copper sulfate) and the H + ions (from water) are attracted to the negative cathode. NCERT Solutions for class 12 Chemistry Chapter 3: Electrochemistry is very essencial for getting good marks in CBSE Board examinations The half-equations for the electrolysis of copper(II) chloride solution (the electrolyte) (a) The negative cathode electrode reaction for the electrolysis of copper(II) chloride solution. You can get ncert solutions and notes for class 12 chapter 3 absolutely free. (iv) An aqueous solution of CuCl2 with platinum electrodes. i.Electrolysis of molten magnesium chloride, MgCl2 Cathode reaction: Mg(2+) + 2e(-) to Mg (Reduction) Anode reaction: 2CI(-) to Cl2 + 2e(-) (Oxidation) ii. (iii) A dilute solution of H2SO4with platinum electrodes. (ii) An aqueous solution of AgNO3with platinum electrodes. (i) An aqueous solution of AgNO3 with silver electrodes. Predict the products of electrolysis in each of the following: Chapter 3: Electrochemistry Chemistry Class 12 solutions are developed for assisting understudies with working on their score and increase knowledge of the subjects. (iv) An aqueous solution of CuCl2 with platinum electrodes. Cyclic voltammetry and potentiostatic electrolysis were used to study the electro-reduction of the Cr 2 O 3-loaded metallic cavity electrode. (iii) A dilute solution of H2SO4with platinum electrodes. The electro-reduction of chromium oxide (Cr 2 O 3) was investigated in an equimolar mixture of CaCl 2 NaCl molten salt at 800 ☌ for developing a more efficient process for chromium preparation. (ii) An aqueous solution of AgNO3with platinum electrodes. The reaction mechanism of electrolytic reduction of SiO2 at a liquid Zn cathode in molten CaCl2 was investigated with the aim of establishing a new production process of solar-grade Si. Question 18: Predict the products of electrolysis in each of the following: (i) An aqueous solution of AgNO3 with silver electrodes. ( Exercise Chapter 3 Electrochemistry Chemistry Class Home UP BOARD Question Papers NCERT Solutions Sample Papers CBSE Notes NCERT Books CBSE Syllabus Predict the Products of Electrolysis in Each of the Following
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
